New Insights Into Dementia with Lewy Bodies, Parkinson’s Disease
Written by |
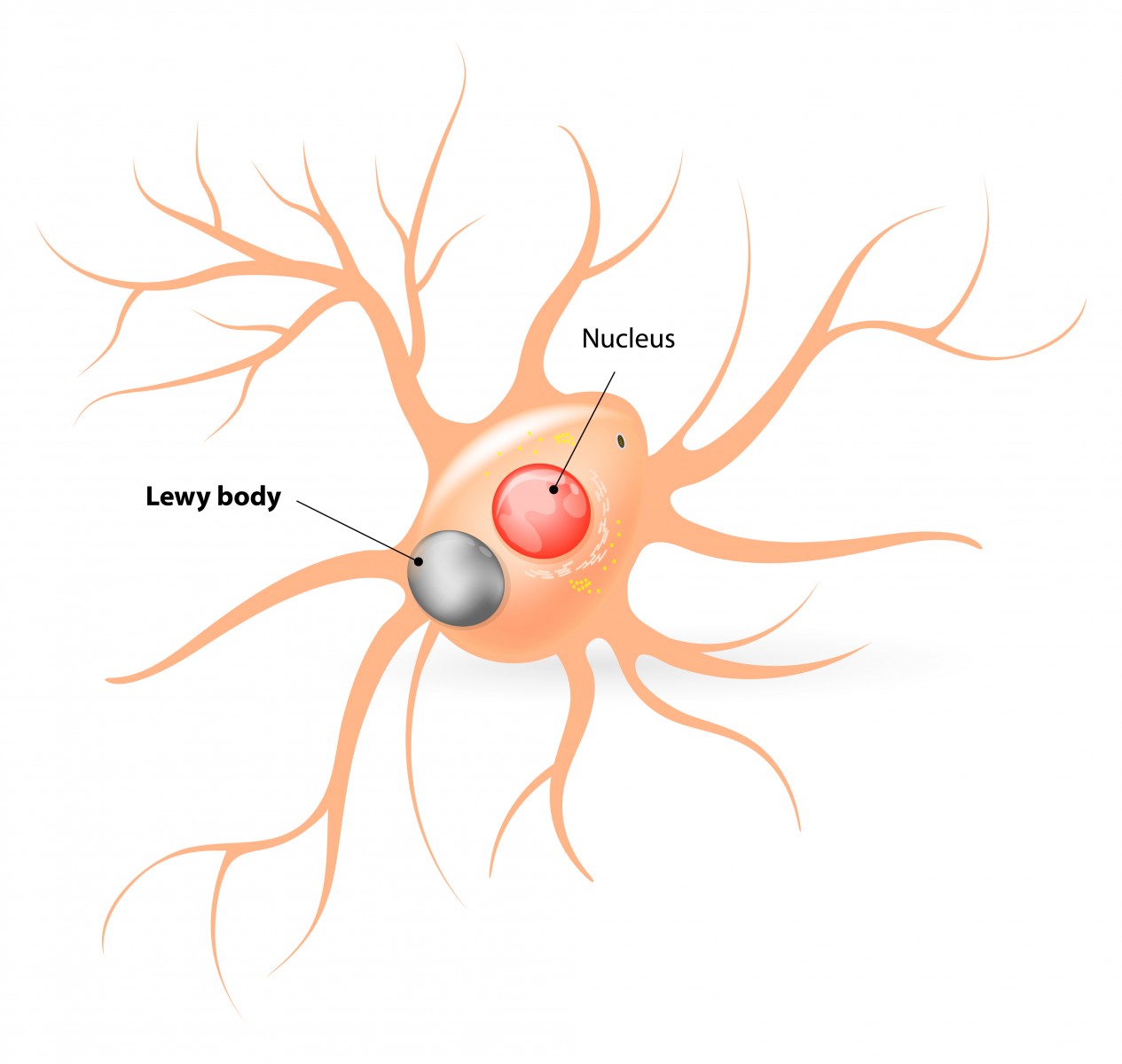
 A new study entitled “Evaluating the relationship between amyloid-β and α-synuclein phosphorylated at Ser129 in dementia with Lewy bodies and Parkinson’s disease” reports a correlation between α-synuclein and amyloid-β in the brain of patients affected by neurodegenerative diseases, such as Parkinson’s disease. The study was published in the journal Alzheimer’s Research &Therapy.
A new study entitled “Evaluating the relationship between amyloid-β and α-synuclein phosphorylated at Ser129 in dementia with Lewy bodies and Parkinson’s disease” reports a correlation between α-synuclein and amyloid-β in the brain of patients affected by neurodegenerative diseases, such as Parkinson’s disease. The study was published in the journal Alzheimer’s Research &Therapy.
In neurodegenerative diseases, including Alzheimer’s and Parkinson’s, Lewy bodies are the most common cause for dementia in affected patients. While Alzheimer’s disease’s key characteristic is the accumulation of protein plaques, constituted by amyloid-β (Aβ), Parkinson’s disease and dementia with Lewy bodies accumulate the protein α-synuclein (α-syn) (with phosphorylation of Serine129 in α-synuclein correlating with disease severity). However, many dementia patients exhibit mixed phenotypes, with Alzheimer’s patients with Lewy body pathology and Parkinson’s disease and dementia with Lewy bodies also exhibiting amyloid-β plaques.
In this study, the authors measured both soluble and insoluble amyloid-β and α-synuclein in human post-mortem brain tissue and analyzed whether there is a relation between the levels of amyloid-β, total α-synuclein, pSer129 α-synuclein and cognitive function ante-mortem in Parkinson’s disease patients. Their main findings were the discovery of a positive correlation between the levels of insoluble pSer129 α-synuclein with insoluble and soluble amyloid-β protein in most brain regions analyzed. The correlation was significantly higher in Parkinson’s disease and dementia with Lewy bodies patients when compared to controls. Additionally, the authors found that the proportion of α-synuclei phosphorylated at Ser129 correlated with ante-mortem mini-mental state examination.
Their findings establish a pathogenic link between the accumulation of amyloid-β and the phosphorylation of Ser129 at α-synuclein, thus increasing disease severity and the probability of developing dementia. The authors highlight that additional studies are required to understand fully how the interaction is maintained.




