ProMIS’s PMN310 Antibody Therapy Binds Only to Toxic Forms of Amyloid Beta, Study Reports
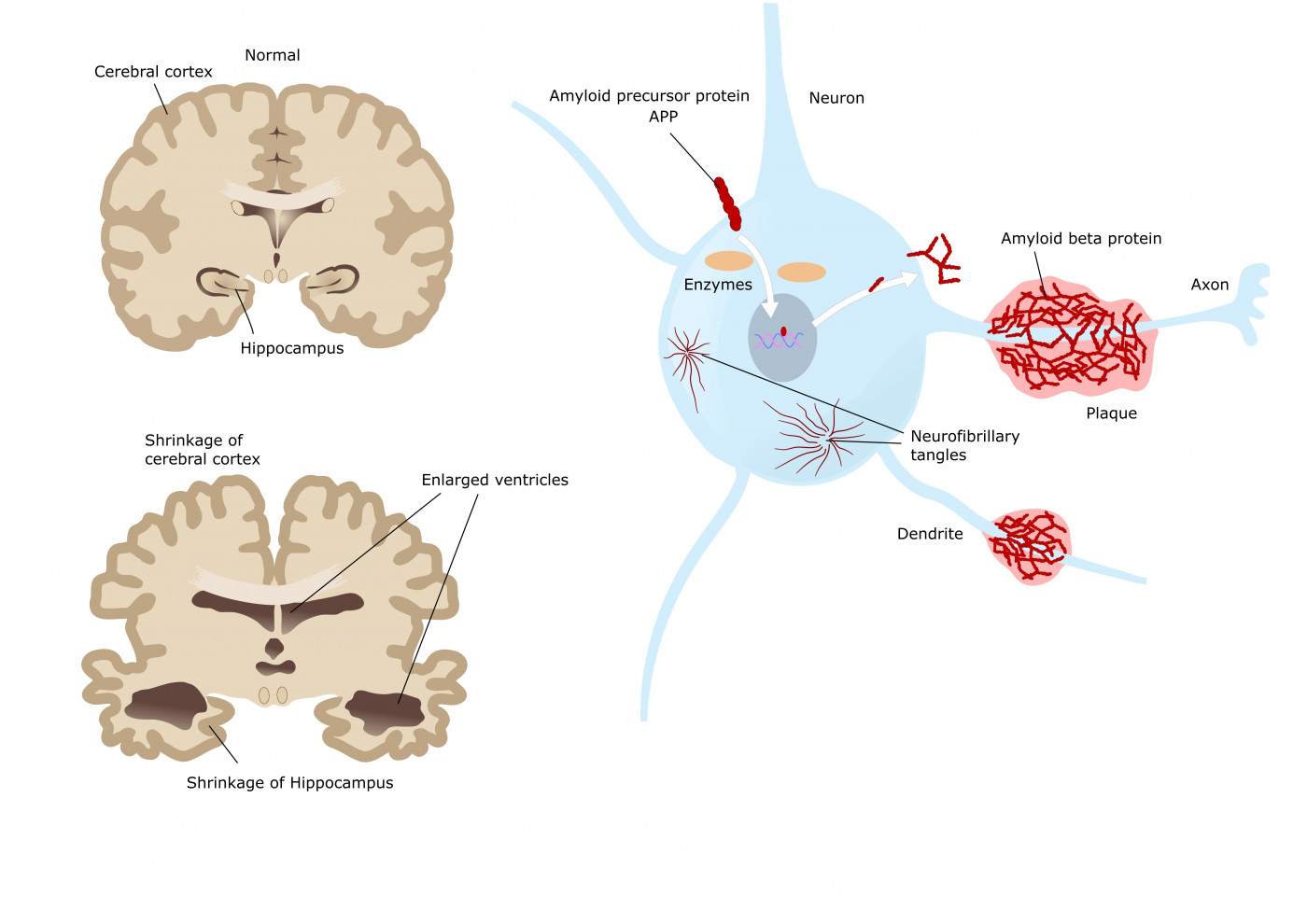
An antibody therapy that ProMIS Neurosciences is developing for Alzheimer’s formed a bond with only the harmful forms of amyloid beta clusters associated with the disease, an indication that it could be an effective treatment, according to a preclinical-trial study.
A good sign was that PMN310 didn’t bind to the forms that are non-toxic, ProMIS said.
The study compared PMN310’s binding characteristics with that of other amyloid beta-directed antibodies for Alzheimer’s. An antibody is a protein that the immune system produces when it detects harmful substances. An antibody therapy involves using an antibody to manage a condition.
“We are pleased to confirm the selective binding of PMN310 to amyloid beta oligomers, a root cause of Alzheimer’s disease,” Dr. Elliot Goldstein, ProMIS’s president and chief executive officer, said in a press release. An oligomer is a molecular complex composed of up to five simpler molecules, or monomers.
“Results of prior clinical studies with other amyloid beta antibody therapeutics clearly showed that antibodies targeting amyloid beta monomer have consistently been ineffective, while those targeting amyloid beta fibrils, the main component of plaque [build-up], are associated with a dose-limiting toxicity of brain swelling,” Goldstein said.
“Taken together, results of prior clinical trials indicate the best-in-class target product profile is selective binding of toxic amyloid beta oligomers” — the approach that PMN310 takes, he said.
ProMIS Neurosciences scientists used a technique called dot blot to compare PMN310’s performance with that of other antibodies targeting amyloid beta. They included Eli Lilly’s solanezumab, Johnson & Johnson and Pfizer’s bapineuzumab, and Biogen’s aducanumab. Bapineuzumab’s production has since been discontinued.
The study showed that PMN310 binds to the toxic form of amyloid beta oligomers, while leaving the other forms untouched. The brain has three forms of amyloid beta. The toxic form that kills nerve cells and promotes neurodegenerative disease is the oligomer form.
In contrast with PMN310, solanezumab binds mainly with amyloid beta monomers, which are harmless, Johanne Kaplan, ProMIS’s chief therapy developer, explained in a webinar. Bapineuzumab can bind to all forms of amyloid beta — monomers, oligomers and fibrils. And aducanumab displays a preference for binding to the toxic oligomer form of amyloid beta and fibrils.
“In conclusion, the antibodies being developed by ProMIS have the potential to selectively target toxic oligomers, thereby avoiding target distraction and side effects,” Kaplan said.






